工藤葵(修士二回生)がポスター賞とAI賞を受賞 / 挑戦的両性花原理 若手の会(2022年10月開催)
工藤葵(修士二回生)が挑戦的両性花原理 若手の会(2022年10月開催)でポスター賞とAI賞を受賞しました。



挑戦的両性花原理 : https://www.ige.tohoku.ac.jp/prg/flower/
Publications リストを更新しました
次の論文をPublicationリストに追加しました。
Chia, K.-S., Kourelis, J., Vickers, M., Sakai, T., Kamoun, S., & Carella, P. (2022). The N-terminal executioner domains of NLR immune receptors are functionally conserved across major plant lineages. BioRxiv, 2022.10.19.512840. https://doi.org/10.1101/2022.10.19.512840 de la Concepcion, J. C., Fujisaki, K., Bentham, A. R., Cruz Mireles, N., Sanchez de Medina Hernandez, V., Shimizu, M., Lawson, D. M., Kamoun, S., Terauchi, R. & Banfield, M. J. (2022). A blast fungus zinc-finger fold effector binds to a hydrophobic pocket in host Exo70 proteins to modulate immune recognition in rice. Proceedings of the National Academy of Sciences, 119(43), e2210559119. https://doi.org/10.1073/pnas.2210559119
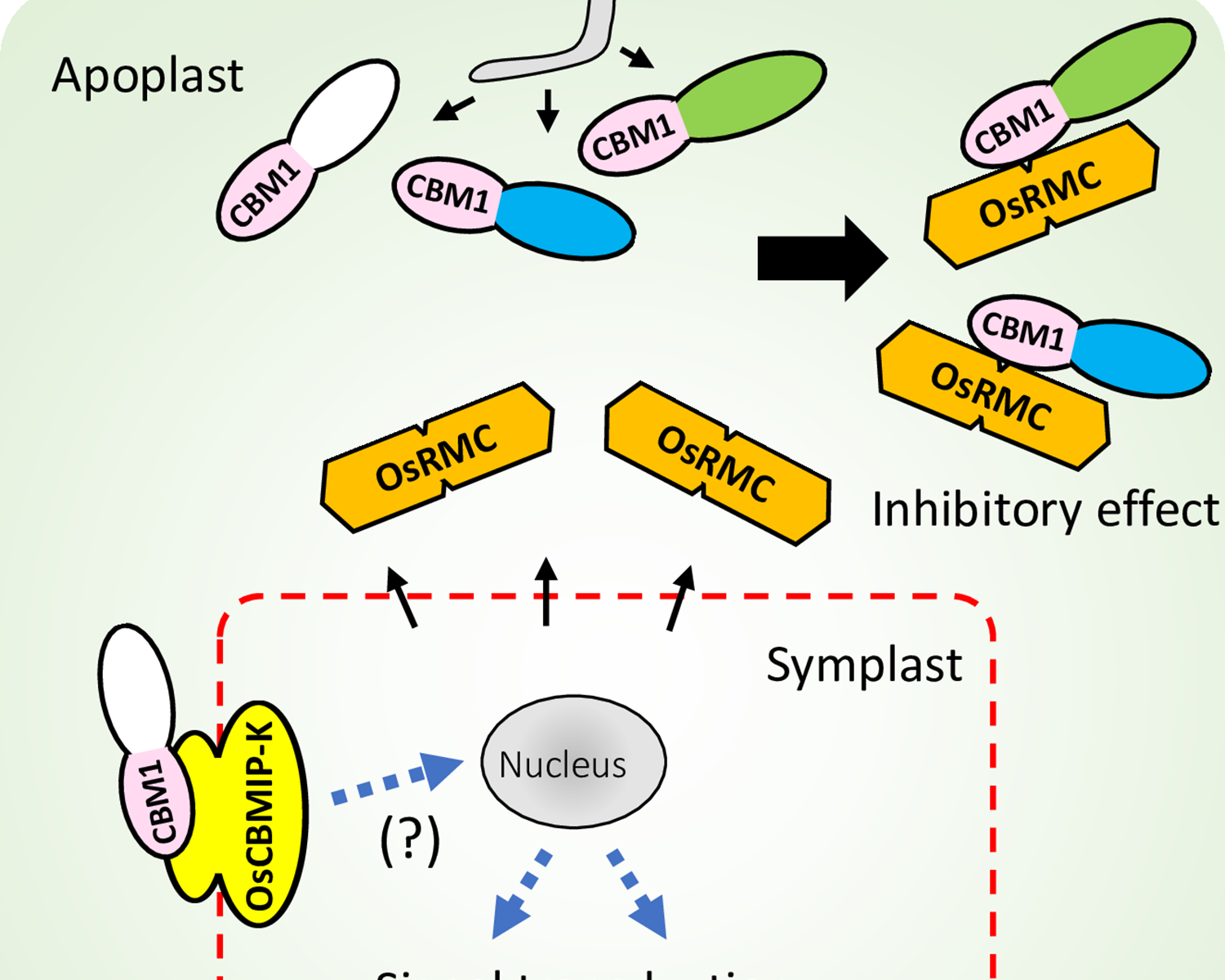
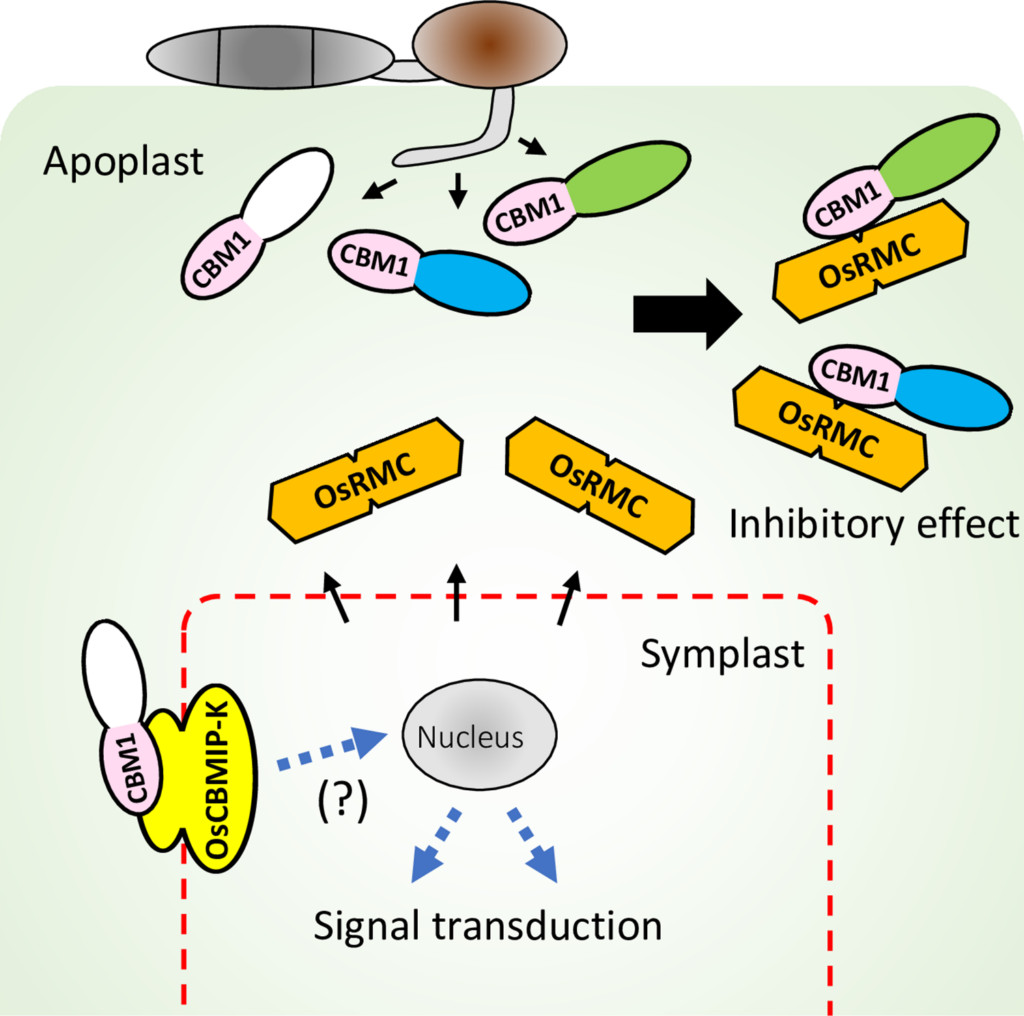
Rice apoplastic CBM1-interacting protein counters blast pathogen invasion by binding conserved carbohydrate binding module 1 motif of fungal proteins
This paper has been published in which Prof. Terauchi is the corresponding author.
Takeda, T., Takahashi, M., Shimizu, M., Sugihara, Y., Yamashita, T., Saitoh, H., Fujisaki, K., Ishikawa, K., Utsushi, H., Kanzaki, E., Sakamoto, Y., Abe, A., & Terauchi, R. (2022). Rice apoplastic CBM1-interacting protein counters blast pathogen invasion by binding conserved carbohydrate binding module 1 motif of fungal proteins. PLOS Pathogens, 18(9), e1010792-. https://doi.org/10.1371/journal.ppat.1010792
Abstract
When infecting plants, fungal pathogens secrete cell wall-degrading enzymes (CWDEs) that break down cellulose and hemicellulose, the primary components of plant cell walls. Some fungal CWDEs contain a unique domain, named the carbohydrate binding module (CBM), that facilitates their access to polysaccharides. However, little is known about how plants counteract pathogen degradation of their cell walls. Here, we show that the rice cysteine-rich repeat secretion protein OsRMC binds to and inhibits xylanase MoCel10A of the blast fungus pathogen Magnaporthe oryzae, interfering with its access to the rice cell wall and degradation of rice xylan. We found binding of OsRMC to various CBM1-containing enzymes, suggesting that it has a general role in inhibiting the action of CBM1. OsRMC is localized to the apoplast, and its expression is strongly induced in leaves infected with M. oryzae. Remarkably, knockdown and overexpression of OsRMC reduced and enhanced rice defense against M. oryzae, respectively, demonstrating that inhibition of CBM1-containing fungal enzymes by OsRMC is crucial for rice defense. We also identified additional CBM-interacting proteins (CBMIPs) from Arabidopsis thaliana and Setaria italica, indicating that a wide range of plants counteract pathogens through this mechanism.
Author summary
Plants have evolved various activity-inhibiting proteins as a defense against fungal cell wall-degrading enzymes (CWDEs), but how plants counteract the function of fungal enzymes containing carbohydrate binding modules (CBMs) remains unknown. Here, we demonstrate that OsRMC, a member of the cysteine-rich repeat secretion protein family, interacts with fungal CBM1. OsRMC binding to CBM1 of a blast fungal xylanase blocks access to cellulose, resulting in the inhibition of xylanase enzymatic activity. Our study provides significant insights into plant countermeasures against CWDEs in the apoplastic space during plant-fungal pathogen interactions. It also reveals a molecular function of the DUF26 domain widely distributed in plant proteins.
(メディア掲載)毎日新聞で当研究室のコムギ遺伝資源保存を取り上げていただきました
2022年8月29日付の毎日新聞夕刊(東京・大阪)およびオンライン版で、当研究室のコムギ遺伝資源の系統保存を取り上げていただきました。
コムギ遺伝資源(種子コレクション)の来歴、系統保存作業の内容、遺伝資源保存の意義、ナショナル・バイオリソース・プロジェクト(NBRP)をわかりやすく紹介してくださっています。
また、農研機構のジーンバンク事業も一緒に紹介されています。
毎日新聞の記事: ウクライナ侵攻で被害の遺伝子銀行 貴重な種子、後世へつなぐ